Avedro: Vedera KXL and Mosaic System
Medical startup Avedro sought to commercialize a device that would reshape the cornea with a low-energy microwave innovation.
Medical startup Avedro sought to commercialize a device that would reshape the cornea with a low-energy microwave innovation.
01
Challenge
LASIK vision-correction surgery is an attractive option for many, but the procedure brings with it certain risks. LASIK involves cutting a flap in the cornea and removing a specific amount of tissue to reshape the corneal surface. The incision is then left to heal itself, increasing the risk of infection and side effects such as glare in low-light conditions.
Medical startup Avedro sought to commercialize a device that would reshape the cornea with a low-energy microwave innovation, one whose procedure did not require cutting into the eye, but offered the same level of vision improvement as LASIK. Once a laboratory prototype of this nascent technology demonstrated the desired results, Avedro needed a more reliable instrument for lab researchers to conduct further tests and gather evidence.
Continuum initially created a high-integrity device with flexible parameters and repeatable output with which Avedro extended their research. When the time came to develop a commercial device, Continuum’s team had the requisite knowledge to take on this task, as they understood the foundational strategies required to develop a sustainable solution. The partnership was extended for the next challenge: productizing the technology and bringing it to market.
03
Solution
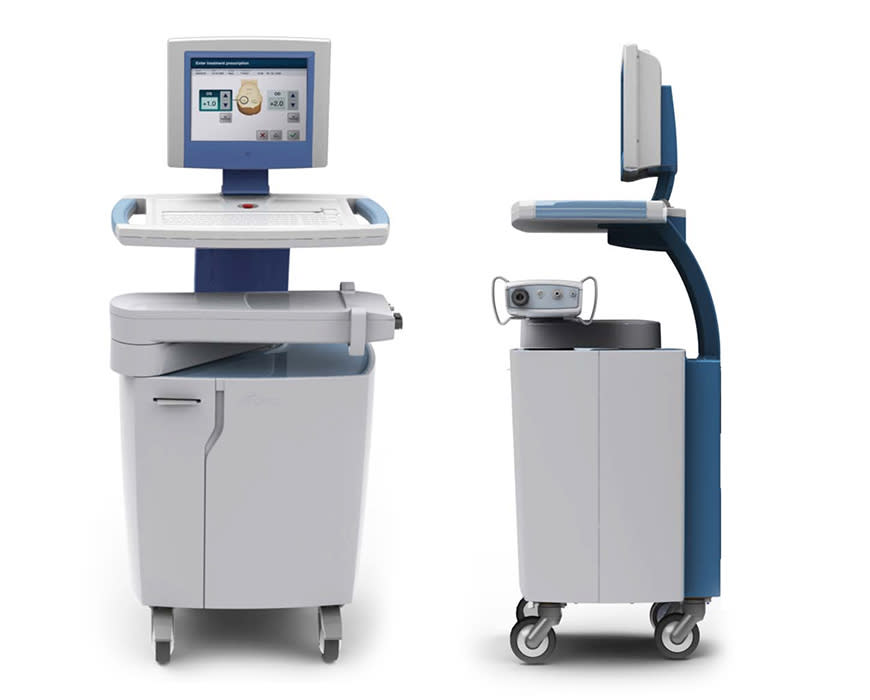
In one year, Continuum designed and developed a fully functional prototype of the Vedera KXS ready for clinical trials. This revolutionary device reshaped the cornea using microwave technology, so the procedure involved no incision or removal of corneal tissue. The benefits of this treatment over LASIK included equivalent vision improvement with immediate results, faster recovery time, and a much lower incidence of side effects.
While the Vedera KXS proved both safe and effective, continued cell growth in the body meant the results of this new procedure could diminish over time. Avedro had a new technology that saturated the cornea with riboflavin molecules. This process increased collagen cross-linking and strengthened the cornea, thereby extending the longevity of the initial procedure’s effects. Because of Continuum’s familiarity with the initial device and deep understanding of the procedure, the team was able to develop this second “cross linking” instrument with ease.
The KXL and Mosaic systems are commercially available in the United States as the first FDA-approved therapeutic treatments for progressive keratoconus and corneal ectasia after refractive surgery (LASIK).





